 Officially, the valence electrons are the electrons in the outer shell ofĮlectrons in its outer shell and so can represent it as a “Cl” with sevenįirst shell and the 8 electrons in the second are inner shell electrons and areĬhart of the main group elements and their Lewis dot symbols. The outer shell are given the name valence electrons. Represent the number of electrons in the outer shell with dots. The third shell can only hold a maximum of 18 electrons but isīecause it is the outer shells that react, we are most interested in the 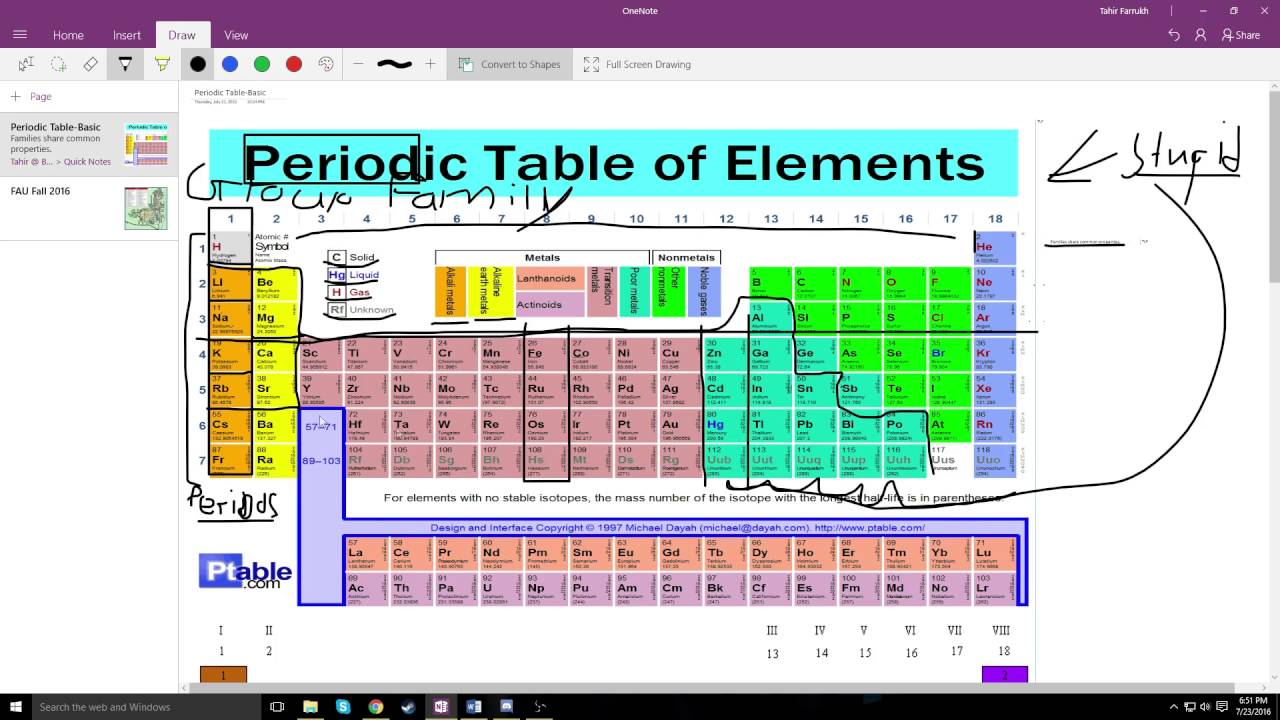
The second shell can only hold a maximum of 8 electrons. The first shell is the smallest so it can only hold a maximum of 2 electrons. Shells correspond to the orbits of the Bohr model. The first two quantum numbers n (the shell) and l (the subshell) are both important in It is the outer shell electrons that form these bonds betweenĪtoms. Identify families of elements and organize research with these effective and efficient graphic organizers.On the left, a periodic table to highlight elements belonging to each family and, on the right, space reserved for notes.May be used in class or for homework, with a variety of resources (textbook, online/e-book). There are some elements that show some, but not all, of the metallicĬalled metalloids and are labeled here are semi-conductors.Įlectrons are the “glue” that hold atoms together in compounds. Nonmetals typically do not show these properties. Have a metallic sheen (shiny) are malleable (bendable) and conduct electricity. The elements can also be divided into two main groups, the metals and the The columns with A (IA through VIIIA) are called the main group 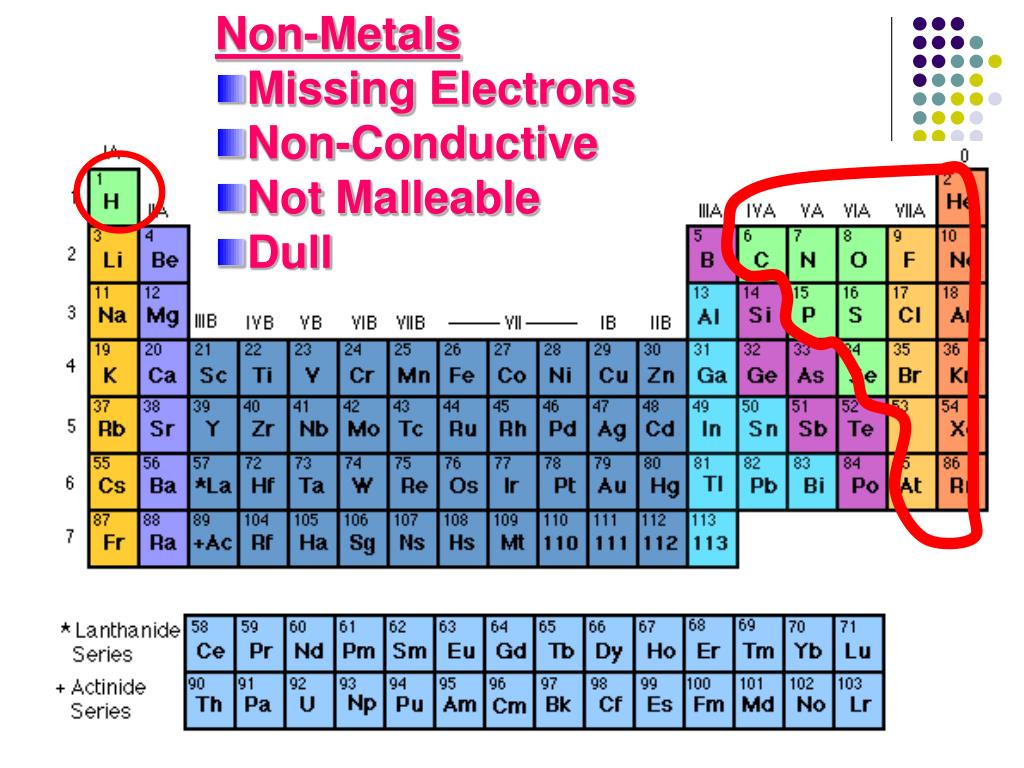
With B (IB through VIIIB) are called the transition elements. Elements with similar reactivity are put into the same column or group. The metals in group IB (copper, silver and gold) are sometimes called the Elements are arranged by reactivity in the periodic table. The elements in group VIIA are called the halogens and theĮlements in group VIIIA are called the noble gases or the inert gases. The elements in group IIA are called the alkaline earth metals. The elements in group IA are called the alkali metals. 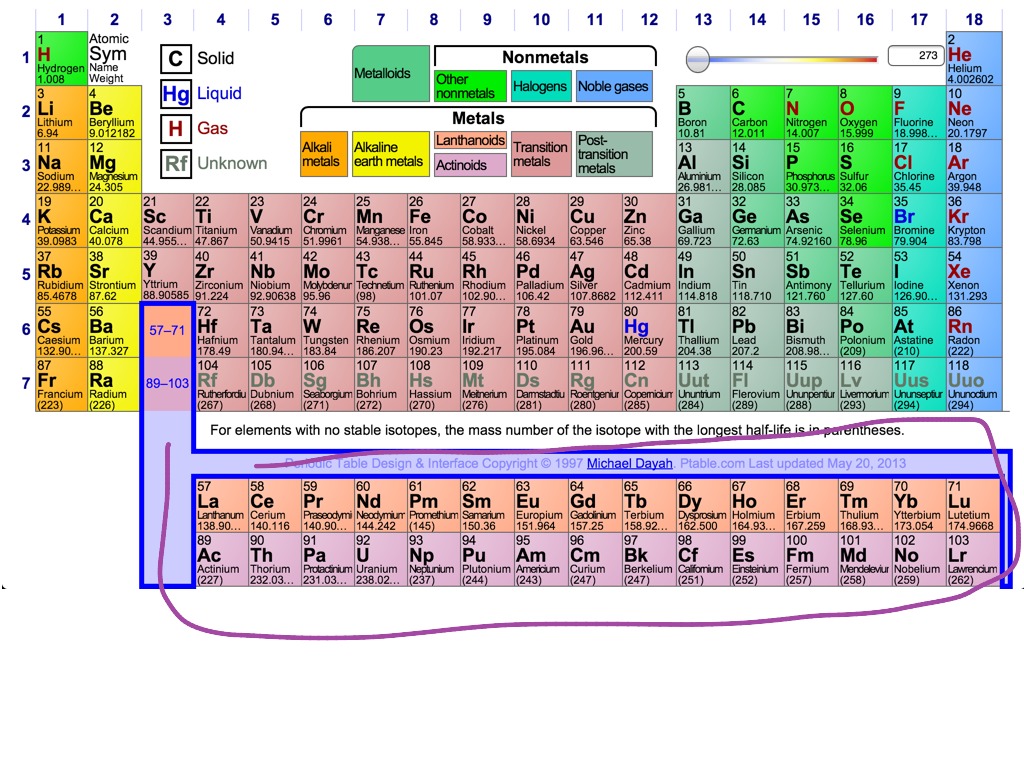
Elements are arranged by reactivity in the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
